ACHAIKI IATRIKI | 2023; 42(1):33–39
Review
Athanasios Moulias, Theoni Theodoropoulou, Angeliki Papageorgiou, Grigorios Tsigkas, Periklis Davlouros
Department of Cardiology, General University Hospital of Patras, Patras 26504, Greece
Received: 11 Apr 2022; Accepted: 20 Jul 2022
Corresponding author: Athanasios Moulias, MD, PhD, Consultant Cardiologist, Department of Cardiology, General University Hospital of Patras, Patras 26504, Greece; Tel.: +302613603281, E-mail: dramoulias@live.com
Key words: Chest pain, acute coronary syndrome, troponin, rapid rule-in, rapid rule-out
Abstract
Chest pain is one of the most common causes of emergency department (ED) visits worldwide. Early diagnosis in patients with suspected myocardial infarction (MI) is of paramount importance, in order to timely provide appropriate therapy and reduce the duration of stay in the ED. For this purpose, high sensitivity cardiac troponin (hs-cTn) assays have been developed and are used by thousands of physicians worldwide. Hs-cTn assays are latest generation tests that allow the detection of very low levels of circulating troponin within a short period of time. When used in the context of established algorithms, hs-cTn measurements reduce the time needed for the safe rule-in or rule-out of MI and, consequently, improve the management of patients presenting with suspected acute coronary syndrome (ACS). However, hs-cTn levels can be elevated in several other conditions associated with cardiomyocyte injury; therefore, the clinician should be aware of the caveats of using rapid rule-in/rule-out algorithms. This article presents the diagnostic utility of the hs-cTn assays and summarizes primary principles for their appropriate, safe and effective use in clinical practice.
INTRODUCTION
It is estimated that as many as 20 million patients present to emergency departments (ED) annually in North America and Europe with various symptoms that may be related to myocardial ischemia [1], such as chest discomfort, shortness of breath, nausea, vomiting, weakness and fatigue. None of these symptoms is specific for acute coronary syndrome (ACS) and, thus, diagnosis is often challenging [2]. In the majority of these patients, the final diagnosis is a noncardiac disorder, rather than ACS, such us pulmonary embolism, pleuritis, chest trauma, acute herpes zoster, rheumatoid arthritis, peptic ulcer or gastroesophageal reflux, etc. [3, 4] (Table 1). Until the development of hs-cTn, the application of a work-up in the ED including clinical assessment (risk factors, symptoms, vital signs), electrocardiogram (ECG) and cardiac biomarkers, resulted in missed MI and inappropriate discharge in ~2% of patients [2]. Missed MI is one of the most frequent malpractices in ED and has great medicolegal consequences. Recently, the use of hs-cTn has decreased this rate and has accelerated the rule-in/rule-out of ACS which is critical for the early initiation of therapy.
Diagnostics
The diagnostic evaluation of a patient presenting in the ED with chest pain (i.e., suspected ACS) should incorporate clinical presentation, physical examination, ECG and hs-cTn measurements in the context of established algorithms.
Clinical Presentation
Acute chest discomfort in ACS patients, usually presents with retrosternal sensation of pain, squeezing pressure or tightness, that may radiate to the left side of the chest, arm, shoulder, neck, jaw, and/or epigastrium and is precipitated by exertion or stress. Clinical presentation may also include accompanying symptoms such as dyspnea, pallor, nausea, vomiting, diaphoresis, anxiety and syncope. Atypical presentations like isolated epigastric pain, isolated dyspnea or minimal chest pain (i.e.,“silent MI”) are more common in older patients, women, diabetics and in patients with chronic renal disease or dementia [4,5].
Physical Examination
Physical examination may be helpful for the differential diagnosis of chest pain (Table 1) in patients with suspected ACS. Findings like heart murmurs, irregular pulse, jugular vein distention, blood pressure difference between upper and lower limbs or between arms and friction rub, may suggest an alternative diagnosis. Additionally, the physical examination may contribute to the distinction between non-coronary causes of chest pain (cardiac tamponade, pericarditis, myocarditis, aortic dissection) and extracardiac pathologies (pneumonia, esophageal perforation, biliary colic, acute pancreatitis) [4].
Electrocardiogram (ECG)
The 12-lead ECG at the ED is the first-line diagnostic tool for the evaluation of patients with suspected ACS and should be performed as soon as possible (within 10 min) of the patient’s arrival. ECG may be normal in more than 30% of patients with Non-ST-segment elevation ACS (NSTE-ACS). However, it may show abnormalities such us ST-segment depression, transient ST-segment elevation or T-wave inversion [4]. Regarding patients with left bundle branch block (LBBB) and a high clinical suspicion of ongoing myocardial ischemia, they should be managed as patients with STEMI irrespective of the time of LBBB appearance [6]. However, hemodynamically stable patients presenting with acute chest discomfort and LBBB have only a slightly higher probability of having MI in comparison with patients without LBBB. Consequently, hs-cTn measurement has a crucial role in deciding whether to perform immediate coronary angiography or not [7].
Standard and High sensitivity cardiac troponin (hs-cTn)
Diagnosis based solely on clinical assessment and ECG seems to be insufficient for patients with suspected NSTE-ACS. Thus, the measurement of a biomarker of cardiomyocyte injury, preferably cTn T or I, is the cornerstone of early diagnosis of MI [4]. Troponins T and I are specific to the heart and are released in the circulation whenever cardiac myocyte damage develops [8].
Recently, the evolution of laboratory techniques has led to the development of new advanced assays, the hs-cTn tests. Hs-cTn tests have a variety of characteristics that differentiate them from the older/conventional troponin test [8]. Firstly, they can detect a much lower serum concentration of cTn with a minimum detection level of 0.005 ng/ml, compared with 0.01 ng/ml when using cTnT (released in 2005) [9]. Moreover, the time frame for the second measurement of hs-cTn can be considerably shortened, due to the rapid detection of any minor myocardial injury [10].
Hs-cTn assays have significant clinical implications compared with standard cTn assays. Most importantly, they have higher negative predictive value for MI and reduce the “troponin-blind” interval leading to earlier diagnosis of MI. Moreover, they have resulted in a ~20% relative increase in the detection of Type I MI and a corresponding decrease in the diagnosis of unstable angina. High sensitivity tests quantify the amount of cardiomyocyte injury [11, 12]. Therefore, they should be interpreted as quantitative variables and not as a binary system (positive/negative). The higher the cTn blood concentration, the higher the probability of MI; elevations up to 3-fold the upper reference limit have only limited (50-60%) positive predictive value (PPV) for MI and may be associated with various conditions. Higher elevations beyond 5-fold the upper reference limit have high (>90%) PPV for acute type 1 MI [13, 14]. The clinician should be aware of the various conditions beyond MI that are commonly associated with an elevation of cTn. These include cardiac conditions such as heart failure, structural heart disease (e.g. aortic stenosis, left ventricular hypertrophy), tachyarrhythmias, hypertensive emergencies, myocarditis, takotsubo syndrome, cardiac contusion, pulmonary embolism, and non-cardiac conditions such as acute neurological events (stroke, subarachnoid hemorrhage), sepsis, etc. [4,12]. It is noteworthy to mention that patients with cTn elevations have a worse prognosis than those with normal levels of cTn, irrespective of the etiology of troponin rise [15].
False positive measurements may be observed in very rare circumstances, in the absence of cardiomyocyte injury. In these cases, heterophilic antibodies or troponin autoantibodies may be present. Thus, if there is inconsistency between clinical presentation and cTn levels, false positive assay results should be considered [16].
Troponin based strategies for rapid rule-in and rule-out of MI
The novel high sensitivity cTn tests have an important clinical advantage; due to their ability to reduce the time interval to the second cardiac troponin assessment, they allow for a rapid diagnosis of MI in the ED. Thus, rapid strategies for the early rule-in and rule-out of MI have been developed and validated in large multicenter studies [14, 17-19]. Two of these strategies, the 0h/1h algorithm (blood draw at 0h and 1h from patient presentation at the ED) and 0h/2h algorithm (blood draw at 0h and 2h), are recommended (Class I, Level of Evidence B) by the European Society of Cardiology (ESC) (Figure 1) [4].
Diagnostic studies validated these two triage algorithms for patients with acute chest pain and/or suspected MI, and defined optimal thresholds for rule-out and rule-in. Sensitivity and negative predictive value (NPV) for MI was found to be equal to 99% and specificity and PPV equal to 70%. The 0h/3h algorithm should be considered as an alternative (Class IIa, Level of Evidence B) [20, 21]. These three strategies (0h/1h, 0h/2h, 0h/3h) are based on the absolute change between two measurements of hs-cTn concentration in the blood. The larger the absolute change of cTn levels within 1h, 2h or 3h, the higher the probability of MI [22]. The cut-off concentrations for the 0h/1h and 0h/2h strategies are assay specific (Table 2) [4]. Clinicians should be aware of the specific assay used in the healthcare facility they are providing service, in order to use the proposed algorithms appropriately.
It is important to mention that these novel strategies detect only MI and not unstable angina (UA). The rapid rule-in/rule-out algorithms should always be used in combination with full clinical assessment and ECG, in order to identify patients at high risk, who are unsuitable for early discharge and need further monitoring. Additional imaging tests, such us echocardiography, stress testing, computed tomography angiography (CTCA) or invasive coronary angiography may be important for an accurate diagnosis. Furthermore, these strategies should only be performed after the exclusion of STEMI from the initial ECG, because these patients need immediate perfusion therapy and so the measurement of cTn is not necessary [4, 12].
0h/1h algorithm
If at presentation (0h) the hs-cTn levels are very low and chest pain onset (CPO) is over 3h, MI can be ruled out. Another occasion where MI can be ruled out is the combination of a low initial concentration of hs-cTn at presentation (0h) and the absence of a significant rise within 1h (No 1hΔ). In cases where the hs-cTn levels at presentation are high or when there is significant increase of hsTn value within the first hour (1hΔ), then the patient is ruled-in for MI [4] (Figure 1).
It is important to highlight that the turnaround time for hs-cTn, in other words the time interval from blood draw until measurements become available to the clinician, is about 1 hour. Therefore, the results from the hs-cTn measurements which are performed at 1 hour after ED presentation will be reported back at about 2 hours after the patient’s arrival at the ED (1h+1h). Thus, the clinicians can make the decision for rule-in or rule-out about 2 to 3 hours after ED presentation [4].
0h/2h algorithm
The 0h/2h strategy measures concentration changes within 2 hours. Absence of significant 2hΔ, means that MI is ruled-out [4].
0h/3h algorithm
The recent ESC guidelines for NSTE-ACS recommend that the 0h/3h algorithm (a rapid rule-out and rule-in protocol with blood sampling at 0 h and 3 h) [23] should be considered as an alternative to the ESC 0h/1h algorithm, if a hs-cTn test with a validated 0 h/3 h algorithm is available [4].
However, evidence suggests that the ESC 0 h/3 h algorithm seems to balance efficacy and safety less well compared with the more rapid protocols (ESC 0h/1h, ESC 0h/2h) [20, 21]
0h/1h vs 0h/3h algorithm
The 0h/1h protocol is preferable in comparison with 0h/3h protocol, due to the fact that the first one allows to rule out more patients than the second one, without an increase in mortality [20]. It is noteworthy to mention that this algorithm has been validated in several multicenter studies and is distinguished for its high efficacy (PPV) and very high safety (NPV). Furthermore, it is simpler to perform, more rapid and is associated with fewer missed MI situations [4, 20].
Observe zone
Patients who cannot be assigned to the rule-out or rule-in zone, are assigned to the ‘observe’ zone. These patients constitute up to one third of those evaluated for suspected ACS and are usually men with pre-existing CAD and high long-term mortality [24]. Additional cardiac troponin measurement at 3 hours and echocardiography are the next steps, crucial for accurate diagnosis [24] (Figure 1).
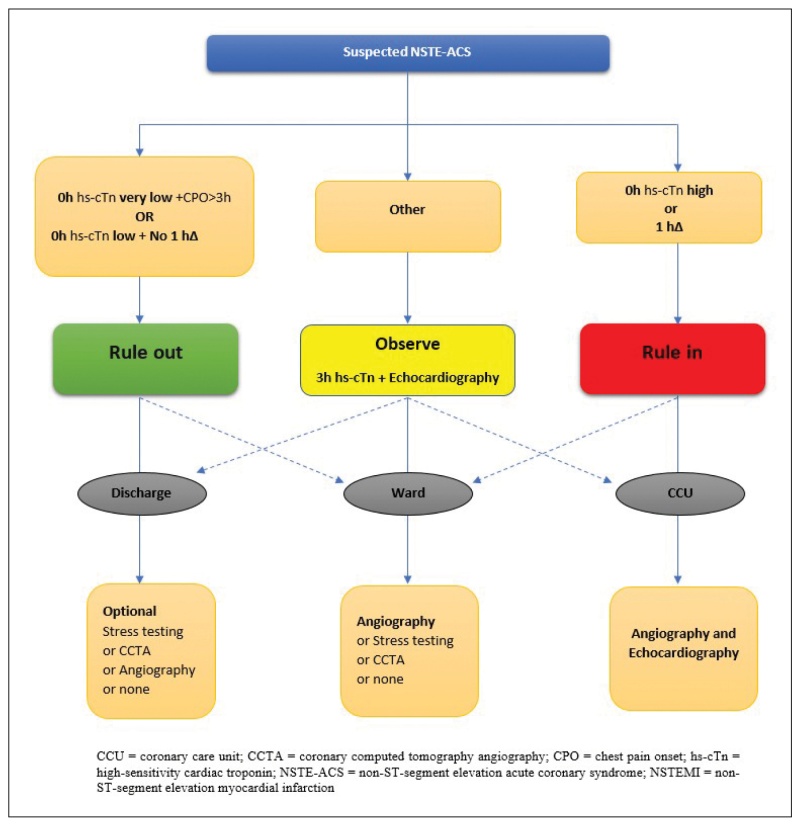
Figure 1. 0 h/1 h rule-out and rule-in algorithm using high-sensitivity cardiac troponin assays in patients presenting with suspected ACS at the emergency department [4].
Clinical assessment of mildly elevated cTn levels is integral, because up to one third of patients assigned to the observe zone will finally have a diagnosis of MI or UA. Thus, serial sampling of cardiac troponin at 3h is essential for the differential diagnosis between acute cardiac disease (MI) and chronic cardiac disease. MI is combined with a dynamic cardiac troponin course, while chronic cardiac disease is associated with a more stable hs-cTn elevation [25].
Patients with a high clinical suspicion of NSTE-ACS and a relevant change of cardiac troponin within 3 hours should undergo invasive coronary angiography, while patients with a low to intermediate suspicion of NSTE-ACS, should be offered noninvasive imaging tests (CCTA) after discharge, or imaging-based stress testing stress echocardiography, positron emission tomography, single-photon emission tomography (SPECT) or cardiovascular magnetic resonance imaging (CMR). In case of special conditions, e.g., rapid ventricular rate response to atrial fibrillation or hypertensive emergency no further diagnostic tests are recommended [4].
Patients with mild hs-cTn elevations
Mildly abnormal hs-cTn levels are just above the 99th percentile (up to 3 times the 99th percentile) and have a broad differential diagnosis [26]. The PPV for patients with acute chest discomfort and mild hs-cTn elevations is very low, about 50% [26]. Therefore, when clinicians are confronted with these challenging patients, they should first consider pre-test probability for MI based on clinical presentation (symptoms and signs) and ECG findings. Moreover, they should think about an obvious non-MI explanation for the mildly abnormal hs-cTn levels, such as acute tachyarrhythmia, acute pulmonary embolism or acute heart failure. They should also consider which diagnostic tests can be useful, such as a repetition of cTn measurement within 1 hour, echocardiography or CMR. Finally, a serious aspect that has to be mentioned, is that hs-cTn elevations, regardless of the cause, are associated with increased mortality. So, further examinations are important [26].
Confounders of cardiac troponin concentration
In patients presenting at the ED with suspected NSTE-ACS, besides the presence or absence of MI, there are four clinical variables that affect hs-cTn levels: age, sex, renal dysfunction, time from chest pain onset [4, 8, 12]
According to recent studies, the use of sex specific cut off levels was associated with an insignificant number of patients being reclassified in comparison with the use of a uniform cutoff level [27, 28]. Consequently, the use of sex-specific cutoff levels is not recommended by ESC so far [4]. Further studies are essential in order to determine the advantages or disadvantages of sex-specific cutoff levels in the diagnostic algorithms.
Patients with suspected MI and renal dysfunction are in a higher risk of MI, than those with a normal kidney function [29]. The diagnosis of MI in these patients is very challenging, because in the first-place patients with renal dysfunction are more prone to an atypical clinical presentation of MI [30]. Moreover, they usually have left ventricular hypertrophy, which can mimic MI findings in ECG. Baseline cardiac troponin concentrations are also chronically elevated in renal dysfunction, in 10-20% of patients for cTn and in up to 70% of patients for hs-cTn, and are associated with poor prognosis [31]. The pathophysiology of high cTn levels is not fully understood, yet.
Even though baseline hs-cTn levels differ between patients with pathological and normal kidney function, there is no difference between them when it comes to measure absolute hs-cTn changes during serial sampling [32].
CONCLUSIONS
Hs-cTn tests combined with clinical evaluation and ECG findings, significantly contribute to the rapid management of patients with suspected MI. Although, these measurements are very useful for the early diagnosis of myocardial infarction, they may also be elevated in several other conditions associated with myocardial injury. Dynamic changes of hs-cTn levels during serial testing are helpful to differentiate ischemic from non-ischemic causes. The most important clinical advantage of hs-cTn assays is the fact that they can be used in the context of novel rapid strategies, allowing for early rule-in and rule-out of MI. Hs-cTn assays not only present a safe, and efficient way for the early detection and management of MI, but they also contribute to a significant reduction of costs and unnecessary investigations in the ED.
Conflict of interest disclosure
None to declare
Declaration of funding sources
None to declare
Author Contributions
Athanasios Moulias, Theoni Theodoropoulou, Angeliki Papageorgiou, Grigorios Tsigkas: drafting of the article; Athanasios Moulias & Periklis Davlouros: critical revision of the article for important intellectual content & final approval of the article.
REFERENCES
1. Blomkalns AL, Gibler WB. Chest pain unit concept: rationale and diagnostic strategies. Cardiol Clin. 2005;23(4):411-21.
2. Pope JH, Aufderheide TP, Ruthazer R, Woolard RH, Feldman JA, Beshansky JR, et al. Missed diagnoses of acute cardiac ischemia in the emergency department. N Engl J Med. 2000;342(16):1163-70.
3. Thygesen K, Mair J, Katus H, Plebani M, Venge P, Collinson P, et al. Recommendations for the use of cardiac troponin measurement in acute cardiac care. Eur Heart J. 2010;31(18):2197-204.
4. Collet JP, Thiele H, Barbato E, Barthelemy O, Bauersachs J, Bhatt DL, et al. 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur Heart J. 2021;42(14):1289-367.
5. Canto JG, Fincher C, Kiefe CI, Allison JJ, Li Q, Funkhouser E, et al. Atypical presentations among Medicare beneficiaries with unstable angina pectoris. Am J Cardiol. 2002;90(3):248-53.
6. Ibanez B, James S, Agewall S, Antunes MJ, Bucciarelli-Ducci C, Bueno H, et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: The Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J. 2018;39(2):119-77.
7. Nestelberger T, Cullen L, Lindahl B, Reichlin T, Greenslade JH, Giannitsis E, et al. Diagnosis of acute myocardial infarction in the presence of left bundle branch block. Heart. 2019;105(20):1559-67.
8. Apple FS, Sandoval Y, Jaffe AS, Ordonez-Llanos J, Bio-Markers ITFoCAoC. Cardiac Troponin Assays: Guide to Understanding Analytical Characteristics and Their Impact on Clinical Care. Clin Chem. 2017;63(1):73-81.
9. Azar RR, Sarkis A, Giannitsis E. A Practical Approach for the Use of High-Sensitivity Cardiac Troponin Assays in the Evaluation of Patients With Chest Pain. Am J Cardiol. 2021;139:1-7.
10. Thygesen K, Mair J, Giannitsis E, Mueller C, Lindahl B, Blankenberg S, et al. How to use high-sensitivity cardiac troponins in acute cardiac care. Eur Heart J. 2012;33(18):2252-7.
11. Reichlin T, Twerenbold R, Reiter M, Steuer S, Bassetti S, Balmelli C, et al. Introduction of high-sensitivity troponin assays: impact on myocardial infarction incidence and prognosis. Am J Med. 2012;125(12):1205-13 e1.
12. Twerenbold R, Boeddinghaus J, Nestelberger T, Wildi K, Rubini Gimenez M, Badertscher P, et al. Clinical Use of High-Sensitivity Cardiac Troponin in Patients With Suspected Myocardial Infarction. J Am Coll Cardiol. 2017;70(8):996-1012.
13. Mueller C. Biomarkers and acute coronary syndromes: an update. Eur Heart J. 2014;35(9):552-6.
14. Neumann JT, Twerenbold R, Ojeda F, Sorensen NA, Chapman AR, Shah ASV, et al. Application of High-Sensitivity Troponin in Suspected Myocardial Infarction. N Engl J Med. 2019;380(26):2529-40.
15. Sorensen NA, Neumann JT, Ojeda F, Giannitsis E, Spanuth E, Blankenberg S, et al. Diagnostic Evaluation of a High-Sensitivity Troponin I Point-of-Care Assay. Clin Chem. 2019;65(12):1592-601.
16. Mair J, Lindahl B, Muller C, Giannitsis E, Huber K, Mockel M, et al. What to do when you question cardiac troponin values. Eur Heart J Acute Cardiovasc Care. 2018;7(6):577-86.
17. Boeddinghaus J, Twerenbold R, Nestelberger T, Badertscher P, Wildi K, Puelacher C, et al. Clinical Validation of a Novel High-Sensitivity Cardiac Troponin I Assay for Early Diagnosis of Acute Myocardial Infarction. Clin Chem. 2018;64(9):1347-60.
18. Neumann JT, Sorensen NA, Schwemer T, Ojeda F, Bourry R, Sciacca V, et al. Diagnosis of Myocardial Infarction Using a High-Sensitivity Troponin I 1-Hour Algorithm. JAMA Cardiol. 2016;1(4):397-404.
19. Twerenbold R, Neumann JT, Sorensen NA, Ojeda F, Karakas M, Boeddinghaus J, et al. Prospective Validation of the 0/1-h Algorithm for Early Diagnosis of Myocardial Infarction. J Am Coll Cardiol. 2018;72(6):620-32.
20. Badertscher P, Boeddinghaus J, Twerenbold R, Nestelberger T, Wildi K, Wussler D, et al. Direct Comparison of the 0/1h and 0/3h Algorithms for Early Rule-Out of Acute Myocardial Infarction. Circulation. 2018;137(23):2536-8.
21. Chapman AR, Anand A, Boeddinghaus J, Ferry AV, Sandeman D, Adamson PD, et al. Comparison of the Efficacy and Safety of Early Rule-Out Pathways for Acute Myocardial Infarction. Circulation. 2017;135(17):1586-96.
22. Mueller M, Biener M, Vafaie M, Doerr S, Keller T, Blankenberg S, et al. Absolute and relative kinetic changes of high-sensitivity cardiac troponin T in acute coronary syndrome and in patients with increased troponin in the absence of acute coronary syndrome. Clin Chem. 2012;58(1):209-18.
23. Wildi K, Nelles B, Twerenbold R, Rubini Gimenez M, Reichlin T, Singeisen H, et al. Safety and efficacy of the 0 h/3 h protocol for rapid rule out of myocardial infarction. Am Heart J. 2016;181:16-25.
24. Nestelberger T, Wildi K, Boeddinghaus J, Twerenbold R, Reichlin T, Gimenez MR, et al. Characterization of the observe zone of the ESC 2015 high-sensitivity cardiac troponin 0h/1h-algorithm for the early diagnosis of acute myocardial infarction. Int J Cardiol. 2016;207:238-45.
25. Lindahl B, Jernberg T, Badertscher P, Boeddinghaus J, Eggers KM, Frick M, et al. An algorithm for rule-in and rule-out of acute myocardial infarction using a novel troponin I assay. Heart. 2017;103(2):125-31.
26. Boeddinghaus J, Reichlin T, Nestelberger T, Twerenbold R, Meili Y, Wildi K, et al. Early diagnosis of acute myocardial infarction in patients with mild elevations of cardiac troponin. Clin Res Cardiol. 2017;106(6):457-67.
27. Mueller-Hennessen M, Lindahl B, Giannitsis E, Biener M, Vafaie M, deFilippi CR, et al. Diagnostic and prognostic implications using age- and gender-specific cut-offs for high-sensitivity cardiac troponin T – Sub-analysis from the TRAPID-AMI study. Int J Cardiol. 2016;209:26-33.
28. Gore MO, Seliger SL, Defilippi CR, Nambi V, Christenson RH, Hashim IA, et al. Age- and sex-dependent upper reference limits for the high-sensitivity cardiac troponin T assay. J Am Coll Cardiol. 2014;63(14):1441-8.
29. Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med. 2004;351(13):1296-305.
30. Aronow WS, Ahn C, Mercando AD, Epstein S. Prevalence of coronary artery disease, complex ventricular arrhythmias, and silent myocardial ischemia and incidence of new coronary events in older persons with chronic renal insufficiency and with normal renal function. Am J Cardiol. 2000;86(10):1142-3, A9.
31. deFilippi CR, Herzog CA. Interpreting Cardiac Biomarkers in the Setting of Chronic Kidney Disease. Clin Chem. 2017;63(1):59-65.
32. Twerenbold R, Wildi K, Jaeger C, Gimenez MR, Reiter M, Reichlin T, et al. Optimal Cutoff Levels of More Sensitive Cardiac Troponin Assays for the Early Diagnosis of Myocardial Infarction in Patients With Renal Dysfunction. Circulation. 2015;131(23):2041-50.